Dipole-dipole forces |
|
|
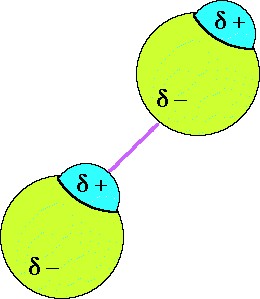
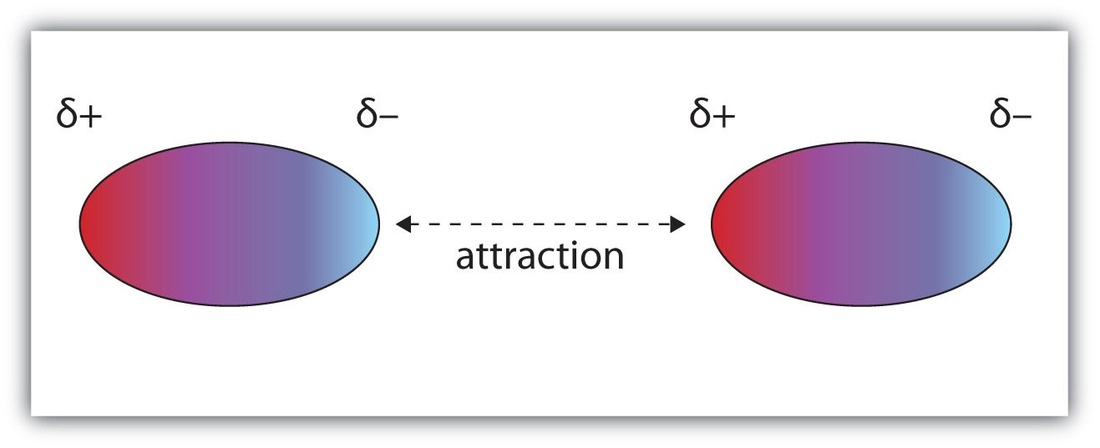
Dipole-dipole forces are attractive forces between the positive end of one polar molecule and the negative end of another polar molecule. They are much weaker than ionic or covalent bonds and have a significant effect only when the molecules involved are close together (touching or almost touching).
In a liquid, the dipole-dipole attraction between molecules become weaker as the moleculers move towards and away from one another. Solid structures are more rigid and those changes are smaller. |
|
When a substance reaches boiling point, these bonds all must be broken in order for the substance to reach a gaseous state.
In short, dipole-dipole forces have the effect of increasing a substance's boiling point due to increasing the amount of energy required to break all its bonds. |